Stem cells from one’s own body fat provide a new treatment option for osteoarthritis, cartilage damage, and sports injuries. Knee joints, hip joints, the joints of the shoulders, hands, and fingers, as well as the spine can be treated. Stem cell therapy is performed on an outpatient basis under local anesthesia. The aim of the treatment is to regenerate and retain the natural joint, relieve pain, and restore mobility.
At a Glance
- Surgery duration: 1 to 3 hours (outpatient)
- Anesthesia: Exclusively local anesthesia (tumescent anesthesia)
- Mobility: Directly after surgery
- Preliminary examinations: Blood test, X-ray/MRI of the joint, further examinations depending on the patiepent’s state of health
- Check-ups: To be arranged on an individual basis
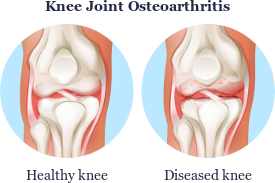
Using Autologous Stem Cells to Treat Osteoarthritis, Deteriorated Cartilages, and Joint Problems
As a consequence of overweight and excessive stress during sports, many people already suffer from joint problems such as cartilage damage, joint wear, osteoarthritis, and injuries at a relatively young age. In most cases, the knee, shoulder and finger joints are affected, and in older persons, also the hip joints and spine.
Apart from weight loss, only physiotherapy, special training or symptomatic pain therapy are recommended for diseases described in medicine as osteoarthritis (also known as arthritis, degenerative arthritis, degenerative joint disease, osteoarthrosis). Subsequently, the progress is observed. If the condition worsens, an artificial joint (joint implant) is inserted.
However, an artificial joint can never replace a natural one. Surgery lasting several hours under general anesthesia is complicated, laborious and expensive. In many cases, blood transfusions are necessary.
Stem Cells Instead of Joint Prosthesis
 The regenerative powers of stem cells have been used in veterinary medicine for some time now. Racehorses and dogs suffering from joint problems and sports injuries are treated with stem cells obtained from the fat of these animals. Stem cells extracted from the animals’ autologous fat appear to be suited for the regeneration of damaged joints, bones and cartilages.
The regenerative powers of stem cells have been used in veterinary medicine for some time now. Racehorses and dogs suffering from joint problems and sports injuries are treated with stem cells obtained from the fat of these animals. Stem cells extracted from the animals’ autologous fat appear to be suited for the regeneration of damaged joints, bones and cartilages.
Results from studies on humans sound promising: Patients treated with stem cells from autologous fat stated that they suffered less pain and that their mobility had increased considerably. If damaged joints are treated early with stem cells, patients could be given a better quality of life and furthermore be spared the necessity of having an artificial joint for the time being.
In my hair salon, I am constantly on my feet. This has put my knees on my knees and I suffered from pain. I wanted to avoid knee surgers and therefore decided to undergo the treatment with fat stem cells. Now I am free of pain and able to ski and bike again. I recommend Clinic DDr. Heinrich®.
 Stem cells and progenitor cells of the connective tissue, so-called mesenchymal stem cells (MSC), may play a pivotal role in the regeneration of deteriorated cartilages, bones and joints. Every person has enough of these vital cells, since fat tissue contains them in large quantities. Artificial reproduction of them in laboratories is therefore unnecessary.
Stem cells and progenitor cells of the connective tissue, so-called mesenchymal stem cells (MSC), may play a pivotal role in the regeneration of deteriorated cartilages, bones and joints. Every person has enough of these vital cells, since fat tissue contains them in large quantities. Artificial reproduction of them in laboratories is therefore unnecessary.
Stem Cell Therapy for Sports Injuries
For torn muscle fibers and also extremely painful sports injuries requiring a longer healing period, there are indications that stem cells have the potential to speed up regeneration and healing.
During treatment, a small quantity of fat is extracted with microcannulas, using local anesthesia, from which the stem cells are then isolated. Immediately afterwards, the stem cells are usually injected into the joint area affected or injured areas, for example, on knees, hips, hands, or fingers. Treatment is carried out on an outpatient basis. The patient is fully mobile afterwards.
 The regenerative effect should begin in the weeks following treatment. A potential repeat treatment session would take place several months after the initial treatment, depending on the extent of symptom improvements.
The regenerative effect should begin in the weeks following treatment. A potential repeat treatment session would take place several months after the initial treatment, depending on the extent of symptom improvements.
Kindly note that no guarantees or promise of cure can be given in medicine generally; this also applies to advanced treatments such as stem cell therapy. Although we carry out your surgery with the greatest care and have achieved excellent results in part with stem cell treatment, adult stem cells are not a universal remedy.
Make appointment or video consultation
Scientific Information
- Aletto, C., Oliva, F., Maffulli, N.: Knee intra-articular administration of stromal vascular fraction obtained from adipose tissue: A systematic review. J Clin Orthop Trauma; 25: 101773. Epub January 22, 2022.
- Arceri, A., Mazzotti, A., Artioli, E., et al.: Adipose-derived stem cells applied to ankle pathologies: a systematic review. Musculoskelet Surg March 2024; 108 (1): 1–9. Epub November 9, 2023. Erratum: Musculoskelet Surg June 2024; 108 (2): 239. Epub April 6, 2024.
- Biazzo, A., D’Ambrosi, R., Masia, F., et al.: Autologous adipose stem cell therapy for knee osteoarthritis: where are we now?. Phys Sportsmed November 2020; 48 (4): 392–9. Epub April 27, 2020.
- Bora, P., Majumdar, A.S.: Adipose tissue-derived stromal vascular fraction in regenerative medicine: a brief review on biology and translation. Stem Cell Res Ther June 15, 2017; 8 (1): 145. Epub June 15, 2017.
- Bui, K.H.-T., Duong, T.D., Nguyen, N.T., et al.: Symptomatic knee osteoarthritis treatment using autologous adipose derived stem cells and platelet-rich plasma: a clinical study. Biomed Res Ther 2014; 1 (1): 2–8. Epub February 7, 2014.
- Chiari, C., Walzer, S., Stelzeneder, D., et al.: Therapeutische Anwendung von Stammzellen in der Orthopädie. Orthopäde December 2017; 46 (12): 1077–90. Epub October 6, 2017. Erratum: Orthopäde Februar 2018; 47 (2): 167. Epub January 5, 2018.
- Davies, B.M., Snelling, S.J.B., Quek, L., et al.: Identifying the optimum source of mesenchymal stem cells for use in knee surgery. J Orthop Res September 2017; 35 (9): 1868–75. Epub March 2, 2017.
- Dubey, N.K., Wei, H.-J., Yu, S.-H., et al.: Adipose-derived Stem Cells Attenuates Diabetic Osteoarthritis via Inhibition of Glycation-mediated Inflammatory Cascade. Aging Dis June 2019; 10 (3): 483–96. Epub June 1, 2019.
- Fodor, P.B., Paulseth, S.G.: Adipose derived stromal cell (ADSC) injections for pain management of osteoarthritis in the human knee joint. Aesthet Surg J February 2016; 36 (2): 229–36. Epub August 3, 2015.
- Freitag, J., Bates, D., Wickham, J., et al.: Adipose-derived mesenchymal stem cell therapy in the treatment of knee osteoarthritis: a randomized controlled trial. Regen Med March 2019; 14 (3): 213–30. Epub February 14, 2019.
- Garza, J.R., Santa Maria, D., Palomera, T., et al.: Use of autologous adipose-derived stromal vascular fraction to treat osteoarthritis of the knee: a feasibility and safety study. J Regen Med March 16, 2015; 4 (1).
- Goncharov, E.N., Koval, O.A., Bezuglov, E.N., et al.: Stromal Vascular Fraction Therapy for Knee Osteoarthritis: A Systematic Review. Medicina (Kaunas); 59 (12): 2090. Epub November 28, 2023.
- Higuchi, J., Yamagami, R., Matsumoto, T., et al.: Associations of clinical outcomes and MRI findings in intra-articular administration of autologous adipose-derived stem cells for knee osteoarthritis. Regen Ther June 2020; 14: 332–40. Epub May 25, 2020.
- Jo, C.H., Lee, Y.G., Shin, W.H., et al.: Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells May 2014; 32 (5): 1254–66. Epub May 17, 2014.
- Kasir, R., Vernekar, V.N., Laurencin, C.T.: Regenerative engineering of cartilage using adipose-derived stem cells. Regen Eng Transl Med December 1, 2015; 1 (1): 42–9. Epub November 30, 2015.
- Kim, K.-I., Kim, M.-S., Kim, J.-H.: Intra-articular Injection of Autologous Adipose-Derived Stem Cells or Stromal Vascular Fractions: Are They Effective for Patients With Knee Osteoarthritis? A Systematic Review With Meta-analysis of Randomized Controlled Trials. Am J Sports Med March 2023; 51 (3): 837–48. Epub January 12, 2022.
- Kim, K.-I., Lee, M.C., Lee, J.H., et al.: Clinical Efficacy and Safety of the Intra-articular Injection of Autologous Adipose-Derived Mesenchymal Stem Cells for Knee Osteoarthritis: A Phase III, Randomized, Double-Blind, Placebo-Controlled Trial. Am J Sports Med July 2023; 51 (9): 2243–53. Epub June 21, 2023.
- Kim, Y.S., Choi, Y.J., Suh, D.S., et al.: Mesenchymal stem cell implantation in osteoarthritic knees: is fibrin glue effective as a scaffold?. Am J Sports Med January 2015; 43 (1): 176–85. Epub October 27, 2014.
- Koh, Y.G., Choi, Y.J., Kwon, O.R., Kim, Y.S.: Second-look arthroscopic evaluation of cartilage lesions after mesenchymal stem cell implantation in osteoarthritic knees. Am J Sports Med July 2014; 42 (7): 1628–37. Epub April 17, 2014.
- Koh, Y.-G., Choi, Y.-J., Kwon, S.-K., et al.: Clinical results and second-look arthroscopic findings after treatment with adipose-derived stem cells for knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc May 2015; 23 (5): 1308–16. Epub December 11, 2013.
- Koh, Y.-G., Choi, Y.-J.: Infrapatellar fat pad-derived mesenchymal stem cell therapy for knee osteoarthritis. Knee December 2012; 19 (6): 902–7. Epub May 14, 2012.
- Koh, Y.-G., Jo, S.-B., Kwon, O.-R., et al.: Mesenchymal stem cell injections improve symptoms of knee osteoarthritis. Arthroscopy April 2013; 29 (4): 748–55. Epub January 29, 2013.
- Koh, Y.-G., Kwon, O.-R., Kim, Y.-S., Choi, Y.-J.: Comparative outcomes of open-wedge high tibial osteotomy with platelet-rich plasma alone or in combination with mesenchymal stem cell treatment: a prospective study. Arthroscopy November 2014; 30 (11): 1453–60. Epub August 16, 2014.
- Krut, Z., Pelled, G., Gazit, D., Gazit, Z.: Stem Cells and Exosomes: New Therapies for Intervertebral Disc Degeneration. Cells; 10 (9): 2241. Epub August 29, 2021.
- Kuroda, R., Ishida, K., Matsumoto, T., et al.: Treatment of a full-thickness articular cartilage defect in the femoral condyle of an athlete with autologous bone-marrow stromal cells. Osteoarthritis Cartilage February 2007; 15 (2): 226–31. Epub September 26, 2006.
- Kyriakidis, T., Iosifidis, M., Michalopoulos, E., et al.: Good mid-term outcomes after adipose-derived culture-expanded mesenchymal stem cells implantation in knee focal cartilage defects. Knee Surg Sports Traumatol Arthrosc February 2020; 28 (2): 502–508. Epub September 6, 2019.
- Lana, J.F.S.D., Lana, A.V.S.D., da Fonseca, L.F., et al.: Stromal Vascular Fraction for Knee Osteoarthritis – An Update. J Stem Cells Regen Med; 18 (1): 11–20. Epub April 5, 2022.
- Lee, H., Lim, Y., Lee, S.-H.: Rapid-acting pain relief in knee osteoarthritis: autologous-cultured adipose-derived mesenchymal stem cells outperform stromal vascular fraction: a systematic review and meta-analysis. Stem Cell Res Ther; 15 (1): 446. Epub November 21, 2024.
- Lee, W.-S., Kim, H.J., Kim, K.I., et al.: Intra-Articular Injection of Autologous Adipose Tissue-Derived Mesenchymal Stem Cells for the Treatment of Knee Osteoarthritis: A Phase IIb, Randomized, Placebo-Controlled Clinical Trial. Stem Cells Transl Med June 2019; 8 (6): 504–11, Epub March 5, 2019.
- Mehling, B., Hric, M., Salatkova, A., et al.: A Retrospective Study of Stromal Vascular Fraction Cell Therapy for Osteoarthritis. J Clin Med Res November 2020; 12 (11): 747–751. Epub November 3, 2020.
- Michalek, J., Moster, R., Lukac, L., et al.: Stromal vascular fraction cells of adipose and connective tissue in people with osteoarthritis: A case control prospective multi-centric non-randomized study.Glob Surg 2017; 3 (3): 1–9. Epub May 29, 2017.
- Michalek, J., Vrablikova, A., Darinskas, A., et al.: Stromal vascular fraction cell therapy for osteoarthritis in elderly: Multicenter case-control study. J Clin Orthop Trauma Januay–Februay 2019; 11 (1): 76–80. Epub November 23, 2018.
- Nguyen, T.A., Hogden, A., Khanna, A., Kuah, D.: Efficacy of adipose-derived stem cells and stromal vascular fraction for pain relief in Kellgren-Lawrence grade II-III knee osteoarthritis: A systematic review (2019–2024). J Orthop December 2025; 70: 95–106. Epub March 21, 2025.
- Ossendorff, R., Menon, A., Schildberg, F.A., et al.: Worldwide Analysis of Adipose-Derived Stem Cells and Stromal Vascular Fraction in Orthopedics: Current Evidence and Applications. J Clin Med; 12 (14): 4719. Epub July 17, 2023.
- Pak, J.: Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose-tissue-derived stem cells: a case series. J Med Case Rep; 5: 296. Epub July 7, 2011.
- Pak, J., Chang, J.-J., Lee, J.H., Lee, S.H.: Safety reporting on implantation of autologous adipose tissue-derived stem cells with platelet-rich plasma into human articular joints. BMC Musculoskelet Disord; 14: 337. Epub December 1, 2013.
- Pak, J., Lee, J.H., Lee, S.H.: A novel biological approach to treat chondromalacia patellae. PLoS One May 2013; 8 (5): e64569. Epub May 20, 2013.
- Pak, J., Lee, J.H., Lee, S.H.: Regenerative repair of damaged meniscus with autologous adipose tissue-derived stem cells. Biomed Res Int; 2014: 436029. Epub January 30, 2014.
- Pers, Y.-M., Rackwitz, L., Ferreira, R., et al.: Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: a phase I dose-escalation trial. Stem Cells Transl Med July 2016; 5 (7): 847–56. Epub May 23, 2016.
- Pintore, A., Notarfrancesco, D., Zara, A., et al.: Intra-articular injection of bone marrow aspirate concentrate (BMAC) or adipose-derived stem cells (ADSCs) for knee osteoarthritis: a prospective comparative clinical trial. J Orthop Surg Res; 18 (1): 350. Epub May 11, 2023.
- Rada, T., Reis, R.L., Gomes, M.E.: Adipose tissue-derived stem cells and their application in bone and cartilage tissue engineering. Tissue Eng Part B Rev June 2009; 15 (2): 113–25.
- Ranmuthu, C.D.S., Ranmuthu, C.K.I., Khan, W.S.: Evaluating the Current Literature on Treatments Containing Adipose-Derived Stem Cells for Osteoarthritis: a Progress Update. Curr Rheumatol Rep; 20 (11): 67. Epub September 10, 2018.
- Richardson, S.M., Kalamegam, G., Pushparaj, P.N., et al.: Mesenchymal stem cells in regenerative medicine: Focus on articular cartilage and intervertebral disc regeneration. Methods. April 15, 2016; 99: 69–80. Epub September 15, 2015.
- Schiavone Panni, A., Vasso, M., Braile, A., et al.: Preliminary results of autologous adipose-derived stem cells in early knee osteoarthritis: identification of a subpopulation with greater response. Int Orthop January 2019; 43 (1): 7–13. Epub October 3, 2018.
- Siennicka, K., Zolocinska, A., Stepien, K. et al.: Adipose-derived cells (stromal vascular fraction) transplanted for orthopedical or neurological purposes: are they safe enough?. Stem Cells Int; 2016: 5762916. Epub September 6, 2016.
- Spasovski, D., Spasovski, V., Bašcarevic, Z., et al.: Intra-articular injection of autologous adipose-derived mesenchymal stem cells in the treatment of knee osteoarthritis. J Gene Med January 2018; 20 (1). Epub January 22, 2018.
- Tangkanjanavelukul, P., Khuangsirikul, S., Heebthamai, D., et al.: Cartilage Regeneration Potential in Early Osteoarthritis of the Knee: A Prospective, Randomized, Open, and Blinded Endpoint Study Comparing Adipose-Derived Mesenchymal Stem Cell (ADSC) Therapy Versus Hyaluronic Acid. In: Int J Mol Sci; 26 (17): 8476. Epub August 31, 2025.
- Vangsness, C.T., Farr, J. II., Boyd, J., et al.: Adult human mesenchymal stem cells delivered via intra-articular injection to the knee following partial medial meniscectomy: a randomized, double-blind, controlled study. J Bone Joint Surg Am January 15, 2014; 96 (2): 90–8.
- Vargel, I., Tuncel, A., Baysal, N., Hartuç-Çevik, I., Korkusuz, F.: Autologous Adipose-Derived Tissue Stromal Vascular Fraction (AD-tSVF) for Knee Osteoarthritis. Int J Mol Sci; 23 (21): 13517. Epub November 4, 2022.
- Wiggers, T.G., Winters, M., Van den Boom, N.A., et al.: Autologous stem cell therapy in knee osteoarthritis: a systematic review of randomised controlled trials. Br J Sports Med October 2021; 55 (20): 1161–9. Epub May 26, 2021.
- Zampogna, B., Parisi, F.R., Ferrini, A., et al.: Safety and efficacy of autologous adipose-derived stem cells for knee osteoarthritis in the elderly population: A systematic review. J Clin Orthop Trauma 2024; 59: 102804. Epub November 7, 2024.
- Zhang, W., Sun, T., Li, Y., et al.: Application of stem cells in the repair of intervertebral disc degeneration. Stem Cell Res Ther; 13 (1): 70. Epub February 11, 2022.
This page serves the purpose of information only and is not to be understood as medical advice. We would like to expressly point out that a cure cannot be guaranteed. Stem cell therapy is an advanced procedure, which has only recently been applied worldwide and for which the long-term studies and reliable documentation on successes, risks and side effects required for a recognized procedure are not yet available.
The legislation on stem cell therapies and conducting of new therapies varies from country to country worldwide. In our clinic in Austria we only carry out the treatments permitted under the applicable legal situation. For other treatments we have worldwide contacts in our cooperation network and can support the selection of specialized medical facilities abroad.








